Kerydin Topical Solution
First FDA-Approved Boron Technology Topical Antifungal Prescription
Kerydin is ONLY Approved for Toenail Infections

- Kerydin (tavaborole) 5% Topical Solution, is the newest FDA-approved TOPICAL antifungal prescription for TOENAIL fungus.
- Kerydin is the first in a new class of prescription antifungal drugs for treatment for toenail fungal infections using Boron technology smart science for effective binding capacity and selectivity for specific fungal targets.
- Kerydin is a SAFE and EFFECTIVE
- No systemic side effects
- Nail the toenail infection from the outside in!
- Simple & Easy to apply
- Available by doctor’s prescription only.
 Kerydin Nail treatment is a 5% solution specifically formulated to reach the site of TOENAIL fungal infections. Kerydin is a clear, quick-drying solution that needs to be applied only once a day. You just use the dropper and apply one or more drops on the top and under the toenail to reach the site of infection. Treat the toenail fungus, dermatophytes, at its source. The active ingredient, tavaborole, is newly FDA approved for TOENAIL fungus infections using Boron technology.
Kerydin Nail treatment is a 5% solution specifically formulated to reach the site of TOENAIL fungal infections. Kerydin is a clear, quick-drying solution that needs to be applied only once a day. You just use the dropper and apply one or more drops on the top and under the toenail to reach the site of infection. Treat the toenail fungus, dermatophytes, at its source. The active ingredient, tavaborole, is newly FDA approved for TOENAIL fungus infections using Boron technology.
Fungal infections of the nails are common and, if left untreated, can progress to a painful debilitating condition. Nail fungal infections are not just a cosmetic thickening and change in shape of the nail, but rather NAIL FUNGUS is a medical condition that is treatable.
Unlike Lamisil tablets that can cause severe systemic side effects like liver failure necessitating a liver transplant, when you can use Kerydin – it’s SAFE and EFFECTIVE. No need for liver function tests.
Nail fungus infection is a very difficult condition to treat. Kerydin toenail fungus solution prescription may be the SAFE antifungal nail treatment you have been searching for. If you have been diagnosed with onychomycosis, toenail fungus infection, Kerydin may be for you.
IMPORTANT WARNING: Do not use Kerydin if you are pregnant or breast feeding or under the age of 18.
IMPORTANT WARNING: Topical Solutions is ONLY approved for use for fungal infections of the TOENAILS.
IMPORTANT WARNING: Do Not use Kerydin in your eyes, mouth or vagina.
IMPORTANT WARNING: Product is flammable – avoid use near heat or open flame
MEDICAL WELLNESS CENTER PROVIDES:
- The easiest, most CONVENIENT, and DISCREET way to receive medical treatment for your most private health needs – All from the PRIVACY and comfort of your home or office! No waiting rooms, No embarrassment, No high fees!
- We DO NOT require you to purchase the medication. Do NOT be fooled by sites that offer free doctor consultation and require medication purchase.
- You can choose most any local pharmacy of your choice to fill your prescription! Choose a pharmacy that accepts your insurance coverage. The prescription cannot be filled at an internet or online pharmacy, or .COM version of a pharmacy, foreign or Canadian pharmacy.
- NO-RISK Online Physician Evaluation. Learn if Kerydin antifungal is for you. If Kerydin is not for you, the visit is FREE.
- If approved, the Physician Consultation fee is only $49.95 plus processing and you will be issued a Kerydin antifungal prescription with refills for 48 weeks of treatment with no further consultation charges.
- If you choose EXPRESS service, your Kerydin prescription will be called in to your local pharmacy within approximately 24 hours.
HOW TO REQUEST A PHYSICIAN CONSULTATION FOR A KERYDIN PRESCRIPTION:
- First read about Kerydin to learn more about its indications and contraindications.
- If you do not have any of the contraindications, next read and agree to the Waiver of Liability.
- Next, accurately and truthfully complete and submit a medical consultation questionnaire.
- Your medical consultation will be reviewed by one of our Physicians. If approved, you will issued a prescription for Kerydin Topical Solution plus refills for a complete 48 week treatment course.
- You can fill the prescription at most any local United States pharmacy of your choice.
- If you desire EXPRESS service, your prescription will be called into a local United States pharmacy of your choice within approximately 24 hours.
- We prescribe Kerydin Topical Solution only for the treatment of mild to moderate fungal infections of the toenail in immunocompetent patients.
- The consultation fee is only $49.95 plus processing. if not approved for Kerydin, there is NO charge.
- If you have any health concerns in any way, you should contact your own physician for further advice.
WHO IS A CANDIDATE FOR Kerydin NAIL PRESCRIPTION?
Nail fungal infections are very common affecting over 30 million Americans. Although, anyone can become infected, fungal nail infections are most common between the ages of 40-65. Those whose nails are exposed to moisture are at a greater risk and some have an inherited tendency to have nail fungal problems. Nail fungus is most commonly seen on the toenails of men and the fingernails of women. Kerydin is only FDA approved for the treatment of TOENAIL infections. Kerydin is NOT approved for fingernail fungal infections.
I If your nails are chipped, hardened, discolored, and abnormally thick sometimes with slight swelling on the skin surrounding the nail, or you have a nail that appears separated from the nail bed, Kerydin Topical Solution is not appropriate for you. If left untreated the condition can progress and other infections may also occur resulting in pain, disfigurement, and disability.
 Normal Healthy Nail Normal Healthy Nail |
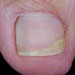 Mild nail fungus infection: 1 or 2 nails involved and less than half the nail shows discoloration Mild nail fungus infection: 1 or 2 nails involved and less than half the nail shows discoloration |
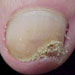 Moderate nail fungus infection: Can involve several nails, more than half of each nail may be thickened and discolored. The nails can be misshaped and can start to split Moderate nail fungus infection: Can involve several nails, more than half of each nail may be thickened and discolored. The nails can be misshaped and can start to split |
 Severe nail fungus infection: Many nails are affected and are brittle, discolored, misshaped, thickened and may start to lift from the nail bed. There is often pain and difficulty using ones fingers or walking. The fungus affects the entire nail including the lunula (the white moon portion at the top part of the nail) with erosion. We do not prescribe Kerydin for severe nail infections! You must seek alternative treatment from you own physician or podiatrist. Severe nail fungus infection: Many nails are affected and are brittle, discolored, misshaped, thickened and may start to lift from the nail bed. There is often pain and difficulty using ones fingers or walking. The fungus affects the entire nail including the lunula (the white moon portion at the top part of the nail) with erosion. We do not prescribe Kerydin for severe nail infections! You must seek alternative treatment from you own physician or podiatrist. |
WHEN TO CONSULT A DOCTOR DIRECTLY RATHER THAN SUBMITTING AN ONLINE CONSULTATION
- If your nail condition has never been diagnosed by a doctor as nail fungus infection (onychomycosis).
- If you have severe nail fungus infection affecting the entire nail including the lunula (the white moon part) with erosion, pictured above, you need to seek direct care from your physician or podiatrist
- We do not prescribe Kerydin if you are HIV-positive, are an organ transplant recipient, have diabetes, diabetic neuropathy, have a long term illness that affects your immune system, compromised immune system, on cancer chemotherapy because you need DIRECT physician supervision
- If you are pregnant or breast-feeding – contact your doctor immediately. You are NOT eligible for an online consultation!
- Kerydin is NOT tested in children under age 18.
Medical Wellness Center’s Online Kerydin Prescription Consultation makes it easy for you to safely and effectively treat toenail fungus infections from the convenience and privacy of your PC without time-consuming and costly doctor visits. If you have any health concerns in any way, you should contact your own physician for further advice.
KERYDIN FAQ
WHAT IS KERYDIN (tavaborole) ANTIFUNGAL TOPICAL NAIL SOLUTION?
Kerydin Nail solution is a NEW, medical-breakthrough. first of a new class of antifungals utilizing Boron technology. Kerydin is a FDA approved TOPICAL nail solution for TOENAIL fungal infections and is active against most strains of Trichophyton rubrum and Trichophyton mentagrophyts. Kerydin topical solution is applied via a dropper, around and under the toenail. It reaches the site of onychomycosis to fight the fungus that lives beneath the nail. The active ingredient in Kerydin (tavaborole) Topical Solution cream is tavaborole 5%, specifically formulated to reach the site of onychomycosis, to treat toenail fungus. Unlike Lamisil tablets nail fungal medications taken orally which can affect the entire body, Kerydin (tavaborole) Topical Solution works from the outside directly on the nail fungus. Lamisil tablets can result in liver failure. Nail the infection from the outside in with Kerydin Topical Nail solution prescription.
WHAT IS A NAIL FUNGUS INFECTION, ONYCHOMYCOSIS?
Onyhomycosis or nail fungus infection is a medical condition that is treatable. Nail fungal infections are caused by dermatophytes, a fungus that infects skin, nails and hair. Kerydin is FDA approved only for the treatment of TOENAIL infections. Most often the fungus enters just under the top of the nail and moves steadily toward the cuticle, the white half moon area. The infected nails become discolored, yellowish-brown, thickened, misshaped and eventually (if left untreated) separate from the nail bed and the nail crumbles off.
WHO IS SUSCEPTIBLE to NAIL FUNGAL INFECTIONS?
Over 30 million Americans are affected. Nail fungus infection is most commonly seen on the toenails of men and the fingernails of women. Most infections occur in those between the ages of 40 to 65 but anyone can become infected. Some individuals have an inherited tendency to have nail fungal problems. Also, individuals with a compromised immune system and certain types of long-term conditions such as HIV-positive, diabetes etc. are at an increased risk. The following are some environmental factors increasing the risk of nail fungal infections: Continuous exposure of hands and feet to moisture, shoes fitting too tightly, not changing shoes often enough, injury to toe or fingernail. Kerydin is FDA approved for the treatment of TOENAIL fungus.
WHY DO I NEED TO TREAT A MILD NAIL INFECTION?
IF LEFT UNTREATED WILL IT GO AWAY BY ITSELF?
Onychomycosis is a progressive fungal infection. The fungus destroys tissue and organic debris accumulates under the nail leading to nail changes beginning with slight thickening and discoloration. If left untreated the nail plate eventually separates from its nail bed and crumbles off and the infections spreads to other nails. The condition progresses from what initially appears to just be a “cosmetic” discoloration of the nail to a painful debilitating condition affecting ones ability to walk or use their fingers.
WHO QUALIFIES FOR AN ONLINE KERYDIN CONSULTATION?
If you have previously been diagnosed by a health care professional as suffering from mild to moderate nail fungal infection (click here for photos of nail fungal infection) you may finally have found a safe and simple solution to the embarrassing condition of discolored, thickened, and hardened nails due to a dermatophyte fungal infection. Kerydin antifungal nail solution is applied directly at the site of the fungal infection and may work for you when other treatments have failed. Kerydin prescription is not for those with severe fungal infections involving the entire nail. Mild to moderate infections are those involving only several nails and more than half of the nail may be affected but NOT including the cuticle white half moon area at the top of the nail where it joins onto the finger. If you have many nails affected, the entire nail is involved, brittle and has lifted off from the nail bed and is accompanied by ulceration of surrounding skin and pain you do NOT qualify for an online consultation, you must seek immediate medical attention from your own physician. If you have long-term conditions affecting your immune system such as diabetes, organ-transplant recipient, HIV-positive, regular user of steroids or steroid inhalers you do not qualify for an online consultation.
WHEN & HOW OFTEN DO I USE KERYDIN (tavaborole) TOPICAL SOLUTION?
 Kerydin Topical Solution is applied only once a day to the top and underside of the toenail of the affected nails with the dropper provided for 48 weeks.
Kerydin Topical Solution is applied only once a day to the top and underside of the toenail of the affected nails with the dropper provided for 48 weeks.
HOW LONG BEFORE I SEE RESULTS?
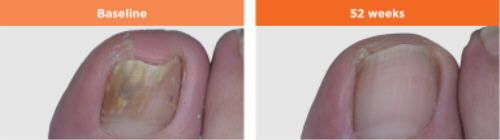
Nail infections are very difficult to cure and it may take many months of treatment to see the effects as toenails grow very slowly. Toenails can take 12 months or longer to completely regrow. Additionally, the time it takes for a healthy nail to grow back varies from person to person. Even after the fungus has been eliminated, nail regrowth can sometimes take a year or longer. But don’t despair and discontinue treatment if you do not see improvement right away. It may take 6 months or longer to start to see improvement. 48 weeks of treatment is required for a cure which is defined as 10% or less residual nail involvement.
HOW IS KERYDIN DIFFERENT FROM THAN OTHER NAIL FUNGAL TREATMENTS?
Nail fungus are very difficult to clear up and if left untreated will only progress. Kerydin (tavaborole) Topical Solution differs from oral medications because it is applied directly to the site of the infected nail. In addition, it does NOT have systemic effects. Since antifungal nail treatment needs to be continued for many months, Kerydin is advantageous as it has no systemic toxicity. Therefore, it is much better tolerated and safer than other available treatments. Kerydin is a NEW FDA-approved topical prescription treatment proven effective for treating mild to moderate TOENAIL fungal infections in immunocompetent candidates. Kerydin is NOT approved for fingernail infections.
HOW DOES KERYDIN DIFFER FROM PENLAC NAIL LACQUER or JUBLIA SOLUTION?
Ciclopirox nail lacquer (Penlac) was the only topical treatment approved by the Food and Drug Administration for the treatment of onychomycosis in the United States, until Jublia solution was FDA approved. Penlac (ciclopirox) nail lacquer has reported complete cure rates of only 5.5-8.5%. In addition, Penlac requires frequent nail debridement and patients have to use alcohol to remove excess buildup of lacquer from the nail plate to avoid additional infection. Jublia has shown complete cure rates of 15-17%. Kerydin (tavaborole) is now the NEWEST antifungal prescription approved by the FDA. Morevere, Kerydin represents the next generation of topical therapies for at-risk patients. Kerydin is the first of its kind utilizing Boron technology. Therefore, it works differently based on its novel antifungal MOA. Most importantly, Kerydin has demonstrated up to 60% clinical improvement.
KERYDIN MECHANISM OF ACTION: Inhibits Protein Synthesis
Tavaborole’s structure contains a boron atom, which enables it to trap tRNA within the editing site of Leucyl-tRNA synthetase (LeuRS).
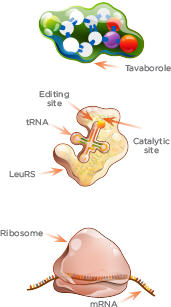 1. LeuRS loads amino acid onto the tRNA at the catalytic site.
1. LeuRS loads amino acid onto the tRNA at the catalytic site.
2. LeuRS checks accuracy of aminoacyl-tRNA at the editing site.
3. Tavaborole targets fungal cytoplasmic LeuRS by binding to the editing site together with tRNA. There, tRNA is trapped.
4. Because tRNA cannot complete the amino acid transfer to the ribosome for assembly, protein synthesis is effectively blocked.
CAN KERYDIN TOPICAL SOLUTION BE USED WITH OTHER ANTIFUNGAL NAIL TREATMENTS OR BE USED WITH NAIL POLISH?
Kerydin Topical solution is NOT recommended to be used with any other systemic antifungal agents for nail fungus infection as there are no studies to determine their interactions. Also DO NOT use nail polish, acrylic nails, or any other cosmetic nail products during treatment regimen with Kerydin. Avoid pedicures, use of nail polish and cosmetic nail products while using Kerydin.
WHAT IF I MISS A KERYDIN (tavaborole) TOPICAL SOLUTION APPLICATION?
If you miss a Kerydin (tavaborole) Topical Solution application it is best to just skip the missed dose and continue on with your daily nail care regimen. Never double-up or use a greater amount to make- up for missed dose, this may only enhance the likelihood of more severe skin irritations.
WHAT ARE THE SIDE EFFECTS OF KERYDIN (tavaborole) TOPICAL SOLUTION?
Kerydin Nail solution does not have systemic effects when used appropriately. There are No know drug interactions and No laboratory monitoring is needed. The majority of patients using Kerydin (tavaborole) Topical Solution experience no side effects, and any side effects are mild if any.
- The most common side effects seen during clinical trials were ingrown toenails, redness, itching, swelling, burning or stinging, blisters, and pain. Kerydin may cause other side effects.
- If nails start to change shape or you have prolonged swelling and pain around your nails, discontinue use and contact your regular doctor.
- If you get the medication in your eyes, rinse immediately with water and seek immediate emergency treatment!
- If you have any signs of an allergic reaction to Kerydin: hives; difficult breathing; swelling of your face, lips tongue, or throat seek immediate emergency medical help.
WHO SHOULD NOT USE KERYDIN NAIL SOLUTION?
-
- Do NOT use Kerydin Topical Solution if you are pregnant or attempting to become pregnant or breast feeding.
- Do NOT use Kerydin on advanced severe fungal nail infections involving the entire nail
- Do NOT use Kerydin on fingernails. Kerydin is FDA-approved for the treatment of TOENAIL fungal infections ONLY!
- We will not prescribe Kerydin Topical Solution to anyone under the age of 18.
-
In order to proceed to the medical consultation, you must go to the next section and read the complete list of contraindications Then you must verify that you do not have any of the contraindications, you are not pregnant and are not breast feeding.
Before Proceeding to the Medical Consultation Form please read
CONTRAINDICATIONS, PRECAUTIONS and WARNINGS
and accept the Waiver of Liability
CONTRAINDICATIONS
Kerydin ANTIFUNGAL Topical Solution is a newly FDA-approved TOPICAL prescription medication for the treatment mild to moderate toenail fungal infections utilizing Boron technology. Kerydin is NOT approved for fingernail fungal infections. Kerydin use is CONTRAINDICATED and must NOT be used in anyone with the following conditions:
- Do NOT use Kerydin if you are pregnant, attempting to become pregnant, or at a high risk of becoming pregnant. Kerydin (tavaborole) is a Pregnancy Kerydin is pregnancy category C, and should not be used during pregnancy.
- Do NOT use Kerydin if you are breast feeding.
- Do NOT use Kerydin if you are allergic or sensitive to any of the ingredients: tavaborole in a solution base alcohol, propylene glycol and edetate calcium disodium.
- Kerydin needs direct supervision if you have a history of immunosuppression or immune system disease, if you are HIV-positive, if you are on cancer chemotherapy treatment, or you have diabetes or diabetic neuropathy.
- Kerydin is contraindicated if you have a severe case of nail fungus infection involving many nails and involving the entire nail to the cuticle with the nail crumbling and lifting from the nail bed accompanied by pain. You need to been seen by your physician or a podiatrist.
- Kerydin is NOT approved for pediatric use, and we do NOT prescribe Kerydin to anyone under the age of 18.
- Kerydin is ONLY approved for use on toenails.
- Do NOT use on fingernails. Do NOT use in eyes, orally, on mucous membranes or in vaginal area.
IMPORTANT PRECAUTIONS and WARNINGS
- Kerydin Topical Solution is for EXTERNAL USE ONLY. Avoid getting Kerydin in your eyes, nose or mouth. Flush with water and seek immediate medical attention!
- Kerydin is for use on nails and surrounding skin only.
- Do NOT use Kerydin in your mouth, eyes, or vagina. Use it exactly as instructed by your physician.
- Apply Kerydin Topical Solution only to toenails.
- Do NOT use on fingernails.
- Do NOT use nail polish, artificial nails, or any other cosmetic nail product on the nails with Kerydin Topical Solution.
- Do NOT combine topical Kerydin Topical Solution with any other systemic antifungal NAIL treatment.
- If the area of application shows signs of increased irritation such as increased redness, swelling, burning, blistering, or oozing, DISCONTINUE USE and seek immediate medical attention from your regular physician.
- Kerydin is to be used as part of a comprehensive nail care program.
- Kerydin topical solution is flammable.
- Keep away from heat and flame. Avoid heat and flame while applying Kerydin to your toenail.
- Keep away from children.
- There are NO systemic effects when used appropriately
- Kerydin is a topically applied solution with negligible systemic absorption and no liver monitoring required
- No need for patient counseling for potential liver toxicity.
- No need for liver function tests
- In vitro liver enzyme studies show that drug interactions are not expected.
SIDE EFFECTS:
The majority of patients using Kerydin Topical Solution experience no side effects. Any side effects are mild, if any. The side effects, when used topically as directed, are limited to:
Adverse events reported by >1% of subjects in Phase 3 studies:Ingrown toenail (2.5% vs 0.3% with vehicle)
Application-site exfoliation (2.7% vs 0.3% with vehicle)
Application-site erythema (1.6% vs 0% with vehicle)
Application-site dermatitis (1.3% vs 0% with vehicle)
IMPORTANT PATIENT INSTRUCTIONS
How to apply Kerydin Topical Solution with its dropper applicator
Kerydin is an oxaborole antifungal dosed once daily for 48 weeks for the treatment of onychomycosis of the toenail(s). Kerydin is specifically formulated to reach the site of the onychomycosis, to treat toenail fungal infections. Kerydin reaches the nail bed and the site of infections when applied as directed. Kerydin antifungal mechanism is via inhibition of protein synthesis.
 Your toenails should be clean and dry before you apply Kerydin
Your toenails should be clean and dry before you apply Kerydin
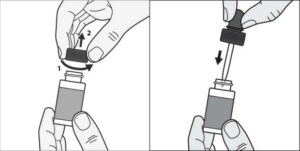
Before you apply Kerydin to your affected toenail for the first time, remove the bottle cap and wrapping from the dropper, and throw it away. Insert the dropper into the Kerydin bottle.

STEP 1: Before you apply Kerydin to your affected toenail, remove the cap from the Kerydin bottle and squeeze to top to get the Kerydin into the dropper.
STEP 2: Hold the dropper over the affected toenail, and apply one or more drops to cover the nail.
STEP 3: Using the dropper tip, spread the Kerydin over the toenail
STEP 4: Apply Kerydin under the toenails. Also use the dropper to spread Kerydin under the entire tip of the toenail.
Allow a couple of minutes for toenails to dry completely.
Repeat steps 1 through 4 for each affected toenail.
Use a tissue to wipe any excess solution from the surrounding skin. Do NOT wipe Kerydin off your toenails.
Step 5: After applying Kerydin to your affected toenails, insert the dropper back into the bottle, screw it on tightly.
Step 6: Wash your hands with soap and water after applying Kerydin.
Sticking With Treatment
Kerydin should be applied once daily for 48 weeks, exactly as directed by your doctor. It may be useful for you to establish a routine or daily treatment time to help you stick to your treatment, such as:
- When you wake up in the morning
- After you shower*
- Just before bed
*Wait for at least 10 minutes after showering, bathing, or washing before applying Kerydin.
I DO NOT HAVE ANY OF THE ABOVE CONTRAINDICATIONS:
In order for Medical Wellness Center Physicians to provide you with the best care, you need to reply honesty to all questions and you need to understand any and all contraindications, risks and side effects associated with Kerydin Topical Solution
By accepting “I Agree” means that you have read the above contraindication, precautions, and warnings and honestly acknowledge that you do not have any of the above contraindications to Kerydin therapy. Also, that I am aware of the side effects! I fully understand that Kerydin is indicated only for mild to moderate fungal infection of the toenails in immunocompetent individuals. Kerydin needs direct patient monitoring in anyone with a compromised immune system such as but not limited to HIV-positive, organ transplant recipient, diabetic, regular use of corticosteroid creams or steroid inhalers. Under conditions of a compromised immune system, fungal infections can progress and with complications can lead to loss of limb or even death. Kerydin is contraindicated in anyone who is pregnant or breastfeeding. I understand that it is solely my responsibility to seek diagnostic and medical treatment for any underlying medical conditions from my own regular primary care physician.
Start Your Online Consult Now!
Where Alternative and Traditional Medicine Come Together for Total Wellness
MEDICAL WELLNESS CENTER: ONLINE HEALTH CARE & PRESCRIPTIONS
(617) 367-8887
wellnessmd@medicalwellnesscenter.com
IMPORTANT NOTE: This is a summary and does not contain all possible information about this product. For complete information about this product or your specific health needs, ask your health care professional. Always seek the advice of your health care professional if you have any questions about this product or your medical condition. This information is not intended as individual medical advice and does not substitute for the knowledge and judgment of your health care professional. This information does not contain any assurances that this product is safe, effective, or appropriate for you.
